CLAIM
1. A method for direct liquefaction of coal, including the following stages:
(1) preparing a coal suspension from raw coal and a catalyst;
(2) mixing the coal suspension with hydrogen and pre-treating the mixture, followed by its supply to the reaction system to carry out the liquefaction reaction;
(3) separation of the reaction products withdrawn from the reactor in a separator (9, 10) with the formation of a liquid phase and a gas phase, while the liquid phase is subjected to fractionation in a distillation column (11) at atmospheric pressure to obtain a product in the form of a diesel fuel fraction and a residual product ;
(4) feeding the residual product obtained in the atmospheric pressure column to a vacuum distillation column (12) to separate into distillate and residue;
(5) mixing the diesel fuel fraction and the distillate to form a mixture and then feeding the mixture into a fluidized bed forced circulation hydroprocessing reactor (13) to carry out the hydrogenation process;
(6) fractionation of the hydrogenation products into petroleum products and a hydrogen donor solvent recycled to step (1).
2. The method according to claim 1, in which step (1) includes the following operations:
(a) converting the raw coal into coal powder of a predetermined particle size after drying and grinding the raw coal in a pre-treatment apparatus; (b) treating the catalyst feedstock (3) and the coal powder in a catalyst preparation apparatus (4) to obtain an ultrafine coal liquefaction catalyst powder; (c) mixing in an apparatus (5) for preparing a suspension of coal liquefaction catalyst and coal powder with a hydrogen-donating solvent (16) to form a coal suspension.
3. The method according to claim 1, in which the stage of the coal liquefaction reaction includes the following operations:
(a) feeding the coal slurry after mixing with hydrogen (6) and preheating into the first fluidized bed reactor (7) with forced circulation to carry out a liquefaction reaction to obtain reaction products exiting the reactor; (b) feeding the reaction products leaving the first fluidized bed reactor (7), after mixing with hydrogen, into a second fluidized bed reactor (8) and forced circulation to continue the liquefaction reaction, said fluidized bed reactors operating at the following conditions: reaction conditions: reaction temperature 430-465°C; reaction pressure 15-19 MPa; the ratio of gas and liquid amounts is 600-1000 nl/kg; volumetric velocity of coal suspension 0.7-1.0 t/m3 h; degree of catalyst addition Fe/dry coal = 0.5-1.0 wt.%.
4. The method according to claim 1, in which step (3) includes the following operations:
(a) feeding the reaction product stream into a high temperature separator (9) for separation into a gas phase and a liquid phase, while the temperature in the high temperature separator is maintained at 420°C;
(b) supplying the gas phase from the high temperature separator (9) to the low temperature separator (10) for further separation into gas and liquid, while the temperature in the low temperature separator is maintained at room temperature.
5. The method according to claim 2, in which -FeOOH is used as a liquefaction catalyst, the particles of which have a diameter of 20-30 nm and a length of 100-180 nm, and the catalyst contains sulfur at a molar ratio of S/Fe=2.
6. The method according to claim 1, in which the hydrogenation at stage (5) is carried out under the following conditions: reaction temperature 330-390°C; reaction pressure 10-15 MPa; the ratio of gas and liquid amounts is 600-1000 nl/kg; flow rate 0.8-2.5 h-1.
7. The method according to claim 1, in which the recirculating hydrogen donor solvent is a hydrogenated liquefied petroleum product with a boiling point in the range of 220-450°C.
8. The method according to claim 1, in which the residue in the vacuum distillation column (12) is characterized by a solid phase content of 50-55 wt.%.
9. The method according to claim 1, in which the mixture of the diesel fuel fraction leaving the atmospheric pressure column and the distillate from the vacuum column has a boiling point of C 5 in the range of 530°C.
10. The method according to claim 1, in which the fluidized bed hydroprocessing reactor (13) with forced circulation is a reactor with internal equipment, wherein a circulation pump is installed near the bottom of the reactor, and the catalyst in the reactor can be replaced during operation.
Synthetic fuel and synthetic oil, new production technology.
Synthetic fuel and synthetic oil can be produced from brown and hard coal using flow-cavitation reactors. This production technology is characterized by high profitability even with small production volumes, environmental friendliness, absence of harmful emissions and low cost of the resulting synthetic fuel.
Synthetic fuel production
Advantages of synthetic fuel
Synthetic fuel is a hydrocarbon fuel that differs from conventional fuel in the production process, that is, obtained by processing a source material that, before processing, has characteristics unsuitable for the consumer.
Synthetic fuels and synthetic oil can be obtained from brown and hard coal. However, the idea of producing liquid motor fuel from coal is not new. Back in the last century, a technology was invented for converting coal into synthesis gas, and then synthesizing petroleum products under pressure based on the so-called Fischer-Tropsch process. Another way is the technology of direct coal liquefaction, developed at the beginning of the 20th century by the German chemist Bergius. But these traditional technologies have not become widespread due to the high cost and bulkiness of the equipment. Basically, such processes were used by states that did not have access to traditional liquid fuel - oil.
In industry, special surfactants (surfactants) can be used, which can be used, for example, for the production of synthetic polymers or the production of emulsions. You can buy surfactants from the manufacturer.
CLAIM
1. A method of burning coal, including drying it, grinding it to a finely dispersed state, mixing the ground coal with a directed oxygen-containing gas flow and combustion, characterized in that the ground coal is heated to a semi-coking temperature of at least 500°C, and volatile gaseous hydrocarbons are released from it, which Then they are separated into liquid and gaseous fractions by condensation, and the semi-coke obtained by heating ground coal is mixed with a directed oxygen-containing gas flow and burned.
2. The method according to claim 1, characterized in that the drying of the ground coal is carried out simultaneously with the grinding of the coal.
3. The method according to claim 1, characterized in that the ground coal is heated to the semi-coking temperature by mixing it with a gaseous coolant.
4. The method according to claim 1, characterized in that the ground coal is heated to the semi-coking temperature by mixing it with a solid coolant having a temperature of 800-1300°C.
5. The method according to claim 3, characterized in that the gaseous coolant is gases formed during the combustion of at least part of the volatile gaseous hydrocarbons.
6. The method according to claim 3, characterized in that the gaseous coolant is gases formed during the combustion of at least part of the resulting charcoal.
7. The method according to claim 4, characterized in that the solid coolant is the resulting semi-coke.
8. The method according to claim 4, characterized in that the solid coolant is quartz sand.
9. The method according to claim 4, characterized in that the solid coolant is ceramic dispersed material.
10. The method according to claim 4, characterized in that the solid coolant is coal.
11. The method according to claim 4, characterized in that the solid coolant is an oxide of an inorganic substance with a fraction size of 0.5-5 mm.
12. The method according to claim 9, or 10, or 12, characterized in that the coolant after use is separated from the char by sifting.
13. The method according to claim 1, characterized in that the gaseous fraction of volatile hydrocarbons is completely or partially burned.
14. The method according to claim 13, characterized in that before combustion, the gaseous fraction of volatile hydrocarbons is purified from sulfur-containing substances.
15. The method according to claim 1, characterized in that the heating of ground coal to the semi-coking temperature is carried out in a vortex chamber by mixing it with hot gas.
Coal liquefaction
Interest in the problem of coal liquefaction is due to the fact that in many cases there is no satisfactory alternative to liquid fuels (such as when using motor fuels). Moreover, the need to increase the use of coal for electricity production puts forward additional requirements for environmental protection, which in turn also increases interest in the problem of obtaining “clean” liquid fuel from coal, which, among other things, is easy to store.
In coals, the ratio of hydrogen and carbon atoms is usually less than one, while in petroleum-type fuels this ratio (depending on the type) ranges from 1.5 to 2. The conversion of coal into liquid fuel requires complex chemical transformations with the introduction into the chemical composition of the resulting product of additional hydrogen and the simultaneous exclusion of oxygen, sulfur and nitrogen from it.
There are three main methods for producing liquid fuel from coal: pyrolysis, hydrogenation and gasification of coal, followed by processing of synthetic gas into liquid fuel using the Fischer-Tropsch method or other technologies. In all of these methods, the presence of hydrogen is necessary: either in the process itself (as in hydrogenation), or subsequently (as in pyrolysis and gasification) to enrich the product initially obtained from coal.
Pyrolysis . In pyrolysis, coal is dry distilled at high temperatures without the presence of air. In this case, through the redistribution of hydrogen in coal, liquid and gaseous products are obtained, as well as a residue in the form of tars or coke, largely freed from hydrogen. The Lurgi-Rurgas pyrolysis method has already been developed for industrial use. Much attention is currently being paid, mainly in the USA and Australia, to the Toscool and COED pyrolysis processes (technological processes that allow obtaining a product with a high content of liquid fractions), on the basis of which the Koolgas process is being developed. Interest in the pyrolysis process is due not only to the fact that it can be used to obtain the maximum amount of liquid products, but also to the possibility of quickly raising the temperature of crushed coal sprayed in the firebox (this property of pyrolysis is used in Germany, Great Britain, the USA and Australia). The main product of all pyrolysis processes is a carbon-enriched product, which can be used directly as fuel and serve as a chemical feedstock for the production of synthetic fuel gas and for the further production of liquid fuel. When processing coals with high sulfur content, the resulting high-carbon product leaves a high level of sulfur content. Liquid derivatives of the pyrolysis process are resins and fuel oils that are complex in their chemical composition, and hydrogenation is required to obtain fuel of the required purity from them.
Hydrogenation. Methods currently being developed for the production of liquid fuel from coal by direct hydrogenation can be divided into two groups - catalytic and non-catalytic methods. There is extensive experience in using both methods in Germany. The industrial application of hydrogenation in the 1920s in the USA made it possible to establish that this process (if carried out under pressure) can significantly increase the quantity and improve the quality of motor fuels when refining crude oil. The first two industrial installations for these purposes were put into operation in the USA in 1930 and 1931. by Standard Oil of New Jersey (now Exom). During the Second World War in Germany, 12 catalytic hydrogenation plants with an annual capacity of 4 million tons were in operation using coal and coal tars using the Bergius-Pierre technology, which accounted for one third of Germany’s total demand for fuel oil, used mainly for motor transport and aviation.
After the war, these installations ceased to exist due to the appearance of cheap petroleum products on the world market. Currently, there are no industrial coal hydrogenation plants. However, intensive research is being carried out in different countries to develop a large number of catalytic and non-catalytic hydrogenation methods.
Non-catalytic processes are essentially designed to directly convert coal in a highly efficient manner into a product that liquefies at high temperatures and minimal hydrogen consumption (hydrogen supply during coal liquefaction is the most expensive procedure). The purpose of this method is to remove carbonaceous formations with a high sulfur content and mineral deposits by physical means and obtain fuel used in the production of electricity and in industrial boilers, or the production of intermediate raw materials, from which, with further enrichment, high-quality motor fuel is obtained.
The most well-developed process is based on the purification of coal by dissolution. An installation using this Solvent Refined Coal (SRC) method with a production capacity of 50 tons/day is currently successfully operating in the USA. There are two variants of this method: SRC-I and SRC-II. When using the SRC-I option, the resulting product is a substance similar to pyrolysis resins at ordinary temperatures, and at a temperature of 170 0C it turns into a liquid. In the liquid phase, this substance is unstable, and given its high viscosity, a serious problem arises in the separation of solid sediment from it. Tightening of legislation regarding the sulfur content in flue gases led to the development of the SRC-II version, in which the process itself is modified: it is carried out at an increased pressure of the supplied hydrogen. In this case, the resulting product remains liquid at normal temperature, the sulfur content in it decreases, but the consumption of hydrogen increases. In 1979, two projects of plants with a consuming capacity of 6000 tons of coal per day were in design study with economic calculations in the United States: one for operation under the SRC-I option and the other under the SRC-II option. Japan and Germany are partners in the SRC-II project. Subvariants of the method of non-catalytic hydrogenation of coal, based on the SRC principle, are actively being studied in Japan, South Africa and the UK. One of the by-products of this method is used in the metallurgical industry, in the production of coke and industrial product with a high carbon content.
The purpose of coal liquefaction using the direct catalytic hydrogenation method is to produce a liquid product that can be directly used as heavy fuel oil or feedstock (synthetic crude oil) and then refined into liquid motor fuels. This process requires the use of catalysts and occurs at significantly higher pressures than with non-catalytic technology.
In the original Bergius technology, extremely high pressure was used (up to 700´105 Pa), and in the latest developments the process takes place under less severe conditions, but the efficiency of the installations has increased significantly and hydrogen consumption has decreased. In the US, the process is carried out using expensive catalysts, which are recovered at the end of the process for regeneration and subsequent recycling; The technology for using cheap catalysts has been developed in Germany.
In the USA, the most developed catalytic hydrogenation method currently being studied is the H-Cole process, in which the problem of controlling the action of the catalyst is solved using a specially designed reactor. This process replicates the H Oil process, which was developed for the upgrading of heavy, high sulfur residual petroleum products. A pilot plant with a capacity of 600 tons of coal per day for the production of fuel oil and 200 tons per day for the production of synthetic crude oil was built in Catlettsburg (West Virginia) and is ready for operation. If the operation of this installation is successful, then it is planned to build a plant with a consuming capacity of 20 thousand tons per day, but completion of this construction is expected at the end of the century.
In Germany, research in this area has reached the stage of industrial testing in a pilot plant with a consuming capacity of 200 t/day. The characteristics of cheap catalysts have made it possible to significantly reduce the operating pressure of the process, and a detailed design of an industrial plant has now begun.
A second method of catalytic hydrogenation that is receiving increased interest is the dissolution process (Exom), the main purpose of which is to avoid the complications caused by direct contact of the catalyst with coal and to separate the liquid product from the solid residue. In this process, the recovered hydrogen solvent is exposed to a catalyst, and the resulting compound reacts with carbon at elevated temperature and pressure. Currently, the construction of a pilot plant with a consuming capacity of 300 tons/day is being completed.
Fischer-Tropsch technology. The Fischer-Tropsch liquid fuel production technology, based on the processing of synthetic gas obtained from coal, was used during the Second World War in Germany. By the end of the war, 9 installations using this technology were operating there, four in Japan, one each in France and Manchuria.
Answers to paragraph 19
1. What are the main natural sources of hydrocarbons that you know? Oil, natural gas, shale, coal.
2. What is the composition of natural gas? Show on a geographical map the most important deposits: a) natural gas; b) oil; c) coal.
3. What advantages does natural gas have over other types of fuel? For what purposes is natural gas used in the chemical industry? Natural gas, compared to other sources of hydrocarbons, is the easiest to produce, transport and process. In the chemical industry, natural gas is used as a source of low molecular weight hydrocarbons.
4. Write the reaction equations for the production of: a) acetylene from methane; b) chloroprene rubber from acetylene; c) carbon tetrachloride from methane.
5. How do associated petroleum gases differ from natural gas? Associated gases are volatile hydrocarbons dissolved in oil. Their isolation occurs by distillation. Unlike natural gas, it can be isolated at any stage of oil field development.
6. Describe the main products obtained from associated petroleum gases. The main products: methane, ethane, propane, n-butane, pentane, isobutane, isopentane, n-hexane, n-heptane, isomers of hexane and heptane.
7. Name the most important petroleum products, indicate their composition and their areas of application.
8. What lubricating oils are used in production? Motor oils, transmission, industrial, lubricating and cooling emulsions for metal-cutting machines, etc.
9. How is oil distilled?
10. What is petroleum cracking? Write an equation for the reactions of hydrocarbon splitting in this process.
11. Why is it possible to obtain no more than 20% gasoline during direct distillation of oil? Because the content of the gasoline fraction in oil is limited.
12. How does thermal cracking differ from catalytic cracking? Give characteristics of thermal and catalytic cracking gasolines. With thermal cracking, it is necessary to heat the reactants to high temperatures, with catalytic cracking, the introduction of a catalyst reduces the activation energy of the reaction, which can significantly reduce the reaction temperature.
13. How can one practically distinguish cracked gasoline from straight-distilled gasoline? Cracked gasoline has a higher octane number compared to straight-distilled gasoline, i.e. is more detonation resistant and is recommended for use in internal combustion engines.
14. What is oil aromatization? Write reaction equations that explain this process.
15. What are the main products obtained from the coking of coal? Naphthalene, anthracene, phenanthrene, phenols and coal oils.
16. How is coke obtained and where is it used? Coke is a solid, gray, porous product obtained by coconut coal at temperatures of 950-1100 without access to oxygen. It is used for smelting cast iron, as a smokeless fuel, a reducing agent for iron ore, and a disintegrant for charge materials.
17. What are the main products obtained: a) from coal tar; b) from tar water; c) from coke oven gas? Where are they used? What organic substances can be obtained from coke oven gas?a) benzene, toluene, naphthalene - chemical industryb) ammonia, phenols, organic acids - chemical industryc) hydrogen, methane, ethylene - fuel.
18. Remember all the main methods for producing aromatic hydrocarbons. What are the differences between the methods for producing aromatic hydrocarbons from the products of coking coal and oil? Write the equations for the corresponding reactions. They differ in the methods of production: primary oil refining is based on the difference in the physical properties of various fractions, and coking is based purely on the chemical properties of coal.
19. Explain how, in the process of solving energy problems in the country, the ways of processing and using natural hydrocarbon resources will be improved. Search for new energy sources, optimization of oil production and refining processes, development of new catalysts to reduce the cost of all production, etc.
20. What are the prospects for producing liquid fuel from coal? In the future, producing liquid fuel from coal is possible, provided that the costs of its production are reduced.
Problem 1. It is known that the gas contains in volume fractions 0.9 methane, 0.05 ethane, 0.03 propane, 0.02 nitrogen. What volume of air will be required to burn 1 m3 of this gas under normal conditions?
Problem 2. What volume of air (no.) is needed to burn 1 kg of heptane?
Task 3. Calculate what volume (in l) and what mass (in kg) of carbon monoxide (IV) will be obtained upon combustion of 5 mol of octane (no.).
We make it from coal
And one more small point that was not mentioned before - when working at home, the installations will probably turn out to be quite large. Therefore, placing them in an apartment is not recommended. But creating them in or near your own home is quite possible.
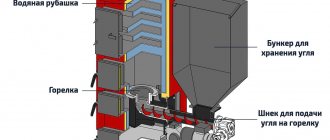
Synthetic gasoline can be produced from coal through the influence of steam. Its gasification is the simplest and most feasible method for home use. So let's get started. Initially, for greater efficiency and increasing the speed of the process, the coal must be crushed. Then it is saturated with hydrogen. Then it is necessary to create a temperature of 400-500 degrees Celsius and a pressure of 50-300 kg/cm2. And we wait for the moment of transition into the liquid state. If a solvent is not used, then only 5-8% of the total mass of coal will become so. Then comes the turn of the catalysts. Suitable for coal: molybdenum, nickel, cobalt, tin, aluminum, iron, and their compounds. Any type of raw material can be used for gasification. Brown, stone - anything will do. Although its quality affects the conversion efficiency. Previously, the designation of the amount of carbon was given and the figure was given as 8%. This is not entirely true. Depending on the brand and quality, the value can range from 4% to 8%. And for minimal suitability for subsequent processing and gasoline recovery, it is necessary to achieve a value of 11% (preferably 15%). Initially, it is not a fact that everything will work out. Especially if you skipped physics and chemistry classes. However, synthetic gasoline can be successfully made and used from coal.
Dynamics of gas consumption from coal in the world
| Intended use | Use in 2001, MW for gas | Share in 2001, % | Commissioned before the end of 2004, MW for gas | Annual capacity increase in 2002-2004, % |
| Chemical production | 18 000 | 45 | 5 000 | 9,3 |
| In-cycle gasification (electricity production) | 12 000 | 30 | 11 200 | 31 |
| Fischer-Tropsch synthesis | 10 000 | 25 | ||
| TOTAL | 40 000 | 100 | 17 200 | 14,3 |
The data presented clearly demonstrate the acceleration of the dynamics of coal gasification involvement in global industry. The increased interest in in-cycle coal gasification in developed countries is explained by two reasons.
Firstly, thermal power plants with intra-cycle gasification are environmentally less hazardous. Gas pre-treatment reduces emissions of sulfur oxides, nitrogen oxides and particulate matter.
Secondly, the use of a binary cycle can significantly increase the efficiency of a power plant and, consequently, reduce specific fuel consumption.
In table Table 2 shows the characteristic values of specific emissions and efficiency for thermal power plants with intra-cycle gasification and for thermal power plants with traditional coal combustion.
table 2
Values of specific emissions and efficiency for thermal power plants with in-cycle gasification and traditional coal combustion
| Options | Traditional coal-fired power plant | TPP with intra-cycle gasification |
| Concentration of harmful substances in flue gases (for coal-fired thermal power plants - according to the European standard), mg/m3 - SOx - NOx - Particulate matter | 130 150 16 | 10 30 10 |
| Electrical efficiency, % | 33-35 | 42-46 |
It should be noted that the specific capital costs when using intra-cycle gasification are approximately 1500 US dollars per 1 kW with the prospect of reducing to 1000-1200 US dollars, while for a traditional coal thermal power plant the specific capital costs are approximately 800-900 US dollars per 1 kW. It is clear that thermal power plants with intra-cycle gasification of solid fuel are more attractive if there are environmental restrictions at the location and when using fairly expensive fuel, since fuel consumption is reduced by 1 kW.
These conditions are typical for developed countries. Currently, the use of intra-cycle gasification of solid fuel is considered the most promising direction in the energy sector.
3.3 Engineering developments over the past century
Currently, the following most cost-effective areas of application of the gasification method have been identified:
— gasification of sulfur and high-ash fuels with subsequent combustion of the resulting gases at powerful thermal power plants. Coals mined annually in Russia contain about 10 million tons of sulfur, most of which, when burned, is released into the atmosphere in the form of toxic sulfur oxides and carbon sulphide. During the gasification of sulfurous coals, hydrogen sulfide is formed, which can be relatively easily extracted and then processed into commercial sulfur or sulfuric acid
— gasification of solid fuels for large-scale production of natural gas substitutes. This direction is of greatest importance for local gas supply to areas remote from natural gas and oil fields or from main pipelines
— gasification of solid fuels to produce synthesis gas, reducing gases and hydrogen for the needs of the chemical, petrochemical and metallurgical industries.
The gasification process depends on many factors that influence the composition of the resulting gas and its calorific value. In this regard, there is still no single generally accepted classification of methods for implementing the process under consideration. Below is one possible classification option.
· by type of blast (gasifying agent): air, air-oxygen, steam-air, steam-oxygen.
· by pressure: at atmospheric pressure, at elevated pressure.
· by fuel particle size: gasification of coarse-grained (lumpy), fine-grained and pulverized fuel.
· according to the design features of the reaction zone: in a stationary dense layer of fuel, in a fluidized layer of fuel, in a pulverized coal torch.
· according to the method of removing ash: in solid form, in the form of liquid slag.
· according to the method of heat supply: with partial combustion of fuel in a gas generator, with mixing of fuel with a preheated solid, liquid or gaseous coolant (regenerative heating), with heat supply through the wall of the apparatus (regenerative heating).
How is gasoline separated from coal?
It is worth noting that the processing of coal in order to obtain different types of motor fuel is not a myth at all. Moreover, there are two proven methods that were put into practice at the beginning of the last century.
In those days, Nazi Germany, striving to conquer all of Europe, provided its military equipment with fuel precisely in these ways, since it did not have its own oil deposits. At the same time, the country had deposits of brown coal, from which synthetic gasoline and diesel fuel were produced at two dozen factories.
For reference. Both methods were invented by different German scientists at the beginning of the 20th century, and accordingly received their names.
As it turns out, coal is not very different in chemical composition from oil. They have a common basis - flammable compounds of carbon with hydrogen, only the proportion of hydrogen in oil is much greater. If the amount of hydrogen in coal can be equalized with oil, then the production of liquid fuel will become a reality. Here are ways to solve the problem:
- hydrogenation, otherwise known as liquefaction (Bergius process).
- gasification followed by fuel synthesis (Fischer–Tropsch process).
To understand whether it will be possible to establish the separation of gasoline at home, you need to get a general understanding of these chemical and technological processes, which will be discussed below.
Carbon monoxide, metal carbonyls and the 18 electron rule
Numerous syntheses based on carbon monoxide and hydrogen are of enormous practical and theoretical interest, since they make it possible to obtain valuable organic compounds from two simple substances. And here the decisive role is played by catalysis by transition metals, which are capable of activating inert molecules CO and H2. Activation of molecules is their transfer to a more reactive state. It should be especially noted that a new type of catalysis has been widely developed in synthesis gas transformations - catalysis by transition metal complexes or metal complex catalysis (see article by O.N. Temkin).
Is the CO molecule really that inert? The idea of the inertness of carbon monoxide is conditional. Back in 1890, Mond obtained from metallic nickel and carbon monoxide the first carbonyl compound of a metal, a volatile liquid with a boiling point of 43°C - Ni(CO)4. The history of this discovery, which can be attributed to chance, is interesting. Mond, studying the causes of rapid corrosion of nickel reactors in the production of soda from NaCl, ammonia and CO2, found that the cause of corrosion was the presence of carbon monoxide impurities in CO2, which reacted with nickel to form tetracarbonyl Ni(CO)4. This discovery allowed Mond to further develop methods for purifying nickel through the production of volatile nickel carbonyl and its subsequent thermal decomposition back to nickel and CO. 25 years later, iron carbonyl, Fe(CO)5, was also accidentally discovered. When BASF opened a long-forgotten steel cylinder containing CO, a yellow liquid was found at the bottom - iron pentacarbonyl, which was gradually formed as a result of the reaction of metallic iron with CO under elevated pressure. Since metal carbonyls are very toxic compounds, at first the attitude of chemists towards them was very cool, but later amazing properties were discovered, including catalytic ones, which determined their widespread use, especially in the chemistry of carbon monoxide. Note that many metals in a finely dispersed state can directly react with carbon monoxide, but only nickel and iron carbonyls are obtained in this way. Carbonyls of other metals are obtained by reducing their compounds in the presence of CO at high pressures.
The composition of transition metal carbonyl complexes can be predicted based on the 18 electron rule, according to which the complex will be stable if the sum of the valence electrons of the metal and the electrons provided by the ligand, in our case CO, is equal to 18, since in this case the electronic configuration corresponds to the stable configuration of the noble atoms gases (krypton).
The carbon monoxide molecule has lone pairs of electrons, and a pair of electrons on the carbon can be provided to form a donor-acceptor bond with the metal. As an example, consider the structure of iron and nickel carbonyls Fe(CO)5 and Ni(CO)4. The iron and nickel atoms have 8 and 10 valence electrons, respectively, and to fill the electron shell of the atom to the configuration of the noble gas atom krypton, 10 and 8 electrons are missing, and therefore, when carbonyls are formed, the iron atom must be provided with electron pairs by five CO molecules, and the nickel atom - four .
Transition metals having an odd number of valence electrons form binuclear carbonyl complexes. Thus, for cobalt, which has nine valence electrons, nine electrons are missing from a stable electronic configuration. Mononuclear complexes, due to the acceptance of four pairs from CO molecules, will have unpaired electrons, and such particles of a radical nature interact with each other to form a metal-metal bond, and as a result, a dimeric complex Co2(CO)8 is formed.
The interaction or coordination of carbon monoxide with a metal leads to a redistribution of electron density not only on CO, but also on the metal, which significantly affects the reactivity of the carbonyl complex. The most common is the so-called linear type of coordination of CO:
In this case, not only the s-interaction occurs due to a free pair of carbon electrons, but also the p-interaction due to the transfer of electrons from the d-orbital of the metal to the energetically accessible vacant orbitals of carbon:
Producing gasoline by gasification
This method, invented by German scientists F. Fischer and G. Tropsch, involves the production of diesel fuel and gasoline by preliminary gasification of coal raw materials. This occurs in a large container - a reactor at temperatures up to 350 ° C and a pressure of no more than 30 Bar. Although the conditions here are not as stringent as for hydrogenation, they are no easier to implement. For example, because superheated water vapor must be blown through a layer of coal under high pressure, which means you cannot do without a powerful steam boiler.
After gasification, the so-called synthesis gas is formed at the outlet of the reactor, consisting of hydrogen and ordinary carbon monoxide (CO). By the way, syngas can be directly used as a gaseous fuel without further processing.
The resulting gases enter the second reactor, where the final processing of coal into liquid fuel occurs. Substances called catalysts are also located there. In industry, different compounds can be used for this purpose, but any of them necessarily contains iron, nickel or cobalt. Without going into the intricacies of chemistry, we note that the output from the second reactor produces fuel, which must still undergo a cracking procedure. That is, the division into gasoline and diesel fuel from coal.
By-products of the reaction are various substances and paraffin. Among the volatile substances released, the largest share is carbon dioxide, which is considered a big problem in the production of fuel using this method. The catalyst also loses activity quite quickly, so it constantly needs to be renewed. These factors, and a number of less significant reasons, lead to the high cost of the product. With an oil price of $50 per barrel, the production of gasoline from coal using the Fischer-Tropsch method is considered unprofitable.
There is another method of coal gasification - thermal. It is similar to the phenomenon of pyrolysis, since it is carried out by heating the raw material in a container from the outside and in the absence of oxygen. Another thing is that the decomposition of solid fuel into gases occurs at a temperature of 1200 ° C, and this requires appropriate equipment. The positive side of the thermal method is that part of the pyrolysis gases is directed to heating the feedstock, and the other to the synthesis of gasoline. Due to this, energy costs are reduced, since coal can heat itself during decomposition.
For reference. On the Internet you can find descriptions of various installations with which you can obtain gasoline from natural gas at home. It is first converted into synthesis gas and then processed into liquid fuel. Even if we assume that these homemade devices are functional, gasifying coal is much more difficult.
Relevance
Thus, there is a need to develop a technology for the primary processing and agglomeration of brown coal that would take into account the specific properties of the original brown coal, the conditions of the process of hydraulic transportation of coal and the requirements for the characteristics of coal raw materials in further processing operations - pyrolysis, combustion, liquefaction, briquetting, dehydration. A solution to this problem can be the technology of treating coal with oil reagents - oil aggregation.
Selective oil aggregation of coal combines a set of processes for structuring a thin polydisperse coal phase in an aqueous environment using oil reagents. The processes of selective oil aggregation of coal are based on the mechanism of adhesive interaction of the oleophilic coal surface with oils, as a result of which its selective wetting and aggregation in a turbulent water flow is achieved. Hydrophilic particles that are not wetted by oil are not included in the structure of the aggregates, which allows them to be isolated in the form of a rock suspension.
Treatment of brown coal with selective oil aggregation eliminates its disintegration and soaking, “preserving” organic matter in hydrophobic aggregates, which are easily dehydrated by mechanical methods and are a good raw material for pyrolysis, briquetting, and gasification.
conclusions
Despite the fact that the separation of motor fuel from hard and brown coal is quite real and has long been tested in production, it is hardly possible to organize it at home. Of course, there will always be a few craftsmen - enthusiasts who love to achieve their goals and will be able to synthesize gasoline with their own hands. But to do this, you need to study the technology in detail and tinker a lot with the equipment, not to mention the fire hazard.
For a wide range of homeowners and car enthusiasts, obtaining diesel fuel and gasoline from coal is not available. And if you approach the issue from an economic point of view, then it is unprofitable. At the moment, until new inventions and developments have appeared on this topic, it is easier and more reliable to use regular, “petroleum” gasoline.
Didn't find the information you are looking for? on our forum.